Ensure Drug Safety & Compliance Today!
Partner with Medivigilax for world-class pharmacovigilance services
Helping Marketing Authorization Holders (MAHs), Pharma Exporters, and Biotech Companies achieve 100% PV compliance, inspection readiness, and operational scalability with or without building internal PV infrastructure.
Due to challenges with worldwide pharmacovigilance legislation, the pharmaceutical business struggled with scattered safety procedures, regulatory non-compliance from limited resources, like high costs and a lack of expertise, making it difficult to meet legal requirements and ensure global safety and compliance.
Medivigilax Research delivers end-to-end pharmacovigilance services, ensuring your products remain compliant throughout their lifecycle with affordable model. We act as an extension of your team, enabling you to focus on growth while we manage your drug safety obligations.

Medivigilax Research Pvt. Ltd. offers comprehensive global Pharmacovigilance and regulatory compliance solutions, including global literature monitoring, regulatory support, training, and Pharmacovigilance audits. Our innovative technology ensures safety and efficacy in pharmaceutical products throughout their lifecycle. We help pharmaceutical manufacturer and biotech firms ensure patient safety, regulatory compliance, and risk management throughout the product lifecycle. Our team of highly qualified professionals ensures compliance with global regulatory authorities.


Medivigilax offers comprehensive pharmacovigilance services to support the entire product lifecycle from clinical trials to post-marketing surveillance.
We establish and maintain a robust pharmacovigilance system tailored to regulatory requirements.
We develop and maintain the PSMF, a critical regulatory document for compliance for medicinal products. Furthermore, It provides an accurate reflection of the entire PV system.
Comprehensive management of adverse event reports from all sources.
Systematic screening of scientific literature for safety signals.
Detection, validation, and evaluation of new or changing safety signals.
Comprehensive preparation and uphold of periodic safety reports like PSUR, PBRER, DSUR and RMP.
Medivigilax Research Private Limited is a leading pharmacovigilance service provider offering comprehensive drug safety and pharmacovigilance outsourcing solutions to pharmaceutical manufacturers who are Marketing Authorization Holders. Our expertise spans ICSR processing, aggregate reporting, Pharmacovigilance System Master File, signal detection, and risk management plans, ensuring full compliance with ICH guidelines, Good Pharmacovigilance Practices, and country specific regulatory guidelines. We deliver cost-effective, scalable, and inspection-ready pharmacovigilance services, helping organizations maintain global regulatory compliance and patient safety throughout the product lifecycle.
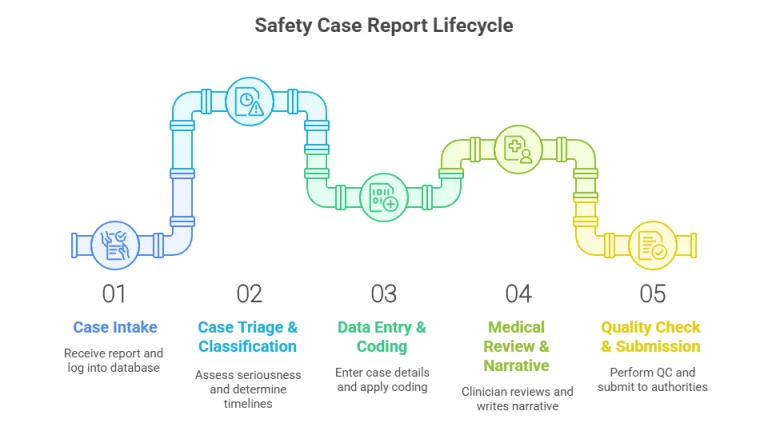
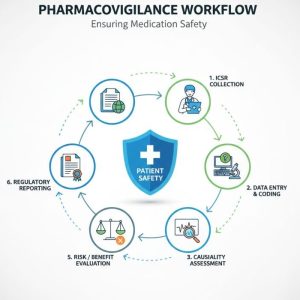
Partner with Medivigilax for world-class pharmacovigilance services
We use cookies to improve your experience on our site. By using our site, you consent to cookies.
Manage your cookie preferences below:
Essential cookies enable basic functions and are necessary for the proper function of the website.
You can find more information in our Home and .